Sickle Cell Program
Building Proposal in support of The Sickle Cell Center of Haiti
To build a Sickle Cell Center on three acres of donated land in St. Marc, Haiti to diagnose and treat patients with sickle cell disease (SCD) and to educate the Haitian medical community, patients with SCD, and their families about the disease and how to best treat it. The Sickle Cell Center of Haiti will dramatically reduce the death rate (estimated at 75% by age 5 years) of babies and children with sickle cell disease and provide jobs to many Haitian people.
Sickle cell disease (SCD) is the most common hemoglobin variant disorder in the world affecting millions of people worldwide. SCD is most common in people of African descent but also affects people in Spanish-speaking regions in the Western Hemisphere (South America, the Caribbean, and Central America); Saudi Arabia; India; and Mediterranean countries such as Turkey, Greece, and Italy.1 The overall prevalence of sickle hemoglobin in Africa ranges from 7-45% depending on the region. By contrast the overall prevalence of the sickle hemoglobin in the US is 1.5% among all births and 7.5% among black births.2 The prevalence of sickle hemoglobin in Haiti is between 8% and 15.2%.3,4
The sickle cell gene is inherited in two severities; sickle cell disease ([SCD] - 2 sickle genes) and sickle cell trait ([SCT] - 1 sickle gene). People with SCD have a life-threatening condition and people with SCT are carriers who live a near normal life. Mortality of African children with SCD before the age of 5 years ranges from 50-90% depending on the economics of the area.5 In comparison, the mortality rate of children born with SCD by age 5 is between 1-2% and falling, reflecting ongoing medical care available to sickle cell patients in the US.1 The prevalence of SCD in Haiti is about 0.6%6 which translates to about 1,500 children born each year with SCD. Based on data from Africa we estimate that approximately 75% of children born with SCD in Haiti die by age 5 years, before they are diagnosed and treated. The Sickle Cell Center of Haiti will change this tragedy.
The alarming death rate in Africa, Haiti, and parts of India is due to a lack of treatment resulting from patients not being diagnosed because of inadequate diagnostic testing methods. Modern, accurate diagnostic testing methods are expensive, require stable electricity, refrigeration, climate control, clean water, and are complicated to perform. Poverty limits patient’s ability to pay for testing which limits the testing methods clinics in underdeveloped countries can make available. Lastly, insufficient education of the lab staff in underdeveloped countries would make the introduction of modern methods difficult even in the absence of financial limitations.
As a result, modern testing is only available in a few, elite clinics in underdeveloped countries while most of the remaining clinics have no testing or offer antiquated, inaccurate testing methods for sickle cell. Therefore, accurate testing for sickle cell is needed that is cheap, fast, and easy, and does not require electricity, clean water, refrigeration, or climate control.
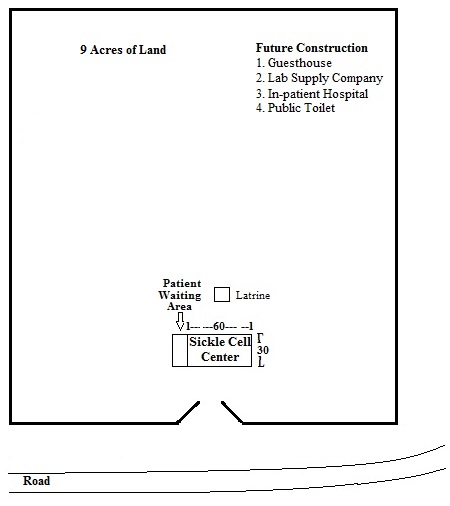
As Founder and President of Randolph World Ministries, Inc. (RWM), a 501c3 Christian-based medical mission ministry, Dr. Randolph plans to establish the first ever Sickle Care Center of Haiti (SiCC). Through our partnership with Partners of the Americas – NJ, RWM has received a donation of land in Saint Marc, Haiti earmarked for the Sickle Care Center of Haiti. Saint Marc is centrally located in Haiti with a major highway and a seaport making it an ideal location for the center.
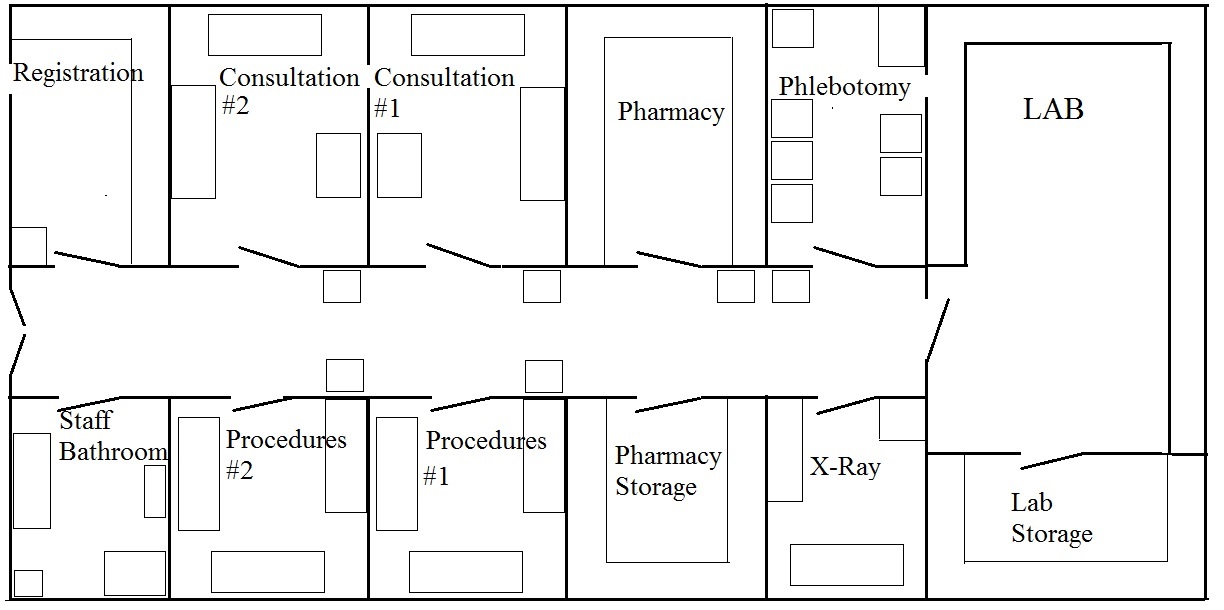
In 2022, the land for the Sickle Care Center (SiCC) was fenced. Construction of the building began in 2023 and the shell of the building was completed in 2024. Unfortunately, progress was halted by political unrest and insecurities in Haiti.
The Sickle Cell Center of Haiti will treat everyone with sickle cell disease regardless of their ability to pay. Therefore, to cover the anticipated deficit in operating costs of the Sickle Cell Center of Haiti, RWM will establish the Laboratory Supply Company of Haiti to sell Sickle Confirm and many other laboratory products. Profits will offset the costs to operate the Sickle Cell Center of Haiti eliminating the need for ongoing, outside financial support once the center is operational.
The Sickle Cell Center of Haiti will reduce the mortality rate of children born with sickle cell disease by delivering: 1. accurate, affordable testing; 2. a high-quality standard of care; and 3. educational programs and consultation services to Haitian healthcare professionals, sickle cell patients, and their families. It will also create jobs for Haitian healthcare professionals (15 to start).
The Laboratory Supply Company of Haiti will: 1. support the sickle cell testing program by distributing Sickle Confirm and other previously unavailable sickle cell testing methods country-wide; 2. decrease operational costs of Haitian clinic clients by selling donated laboratory supplies and other laboratory products at wholesale prices; 3. expand laboratory offerings of Haitian clinic clients by offering non-sickle cell related testing materials not previously available in Haiti; and 4. create jobs (2 jobs/Laboratory Supply Company branch to start).
We see the plan for the SiCC unfolding in four major phases:
Phase 1: Construction of the Sickle Care Center of Haiti with simultaneous opening of one location of the Laboratory Supply Company of Haiti (Year 1-2);
Phase 2: Operating the Sickle Care Center of Haiti and the Laboratory Supply Company of Haiti (Year 3);
Phase 3: Construction of ancillary structures (Years 4-5) on Sickle Care Center of Haiti property to include:
a. In-patient rooms for admitted patients – attached to the SiCC;
b. Guesthouse for medical teams – separate building;
c. public toilet – separate building;
Phase 4: Opening additional Laboratory Supply Companies as needed to keep pace with growth of the sickle cell program (>5 years).
Once we have documented that this model will reduce the mortality rate of sickle cell disease and show financial sustainability without outside support, our vision is to replicate this model in sub-Saharan African countries and the rest of the world to reduce the sickle cell tragedy worldwide.
-
Centers for Disease Control (CDC) https://www.cdc.gov/ncbddd/sicklecell/data.html
-
Ojodu J, Hulihan MM, Pope SN, Grant A. Incidence of Sickle Cell Trait — United States, 2010. Morbidity and Mortality Weekly. December 12, 2014 / 63(49);1155-1158.
-
Carter TE, von Fricken M, Romain JR, Memnon G, Victor YS, Schick L, Okech BA, and Milligan CJ. Detection of Sickle cell Hemoglobin in Haiti by Genotyping and Hemoglobin Solubility Tests. Am J Trop Med Hyg. 2014;91(2):406-411.
-
Randolph TR. Estimated Prevalence of Sickle Cell in Northern Haiti. Clin Lab Sci. 2010;23(2):79-83.
-
Grosse SD, Odame I, Atrash HK, Amendah DD, Piel FB, and Williams TN. Sickle Cell Disease in Africa - A Neglected Cause of Early Childhood Mortality. Am J Prev Med. 2011 Dec; 41(6): S398–S405.
-
Fleur RS, Archer N, Hustace T, et. al. Capacity Building and Networking to Make Newborn Screening for Sickle Cell Disease a Reality in Haiti. Blood Advances. 2018 Nov;2(1):54-55.
-
Randolph TR and Wheelhouse J. Novel Test method (Sickle Confirm) to Differentiate Sickle cell Anemia from Sickle Cell Trait for Potential Use in Developing Countries. Clin Lab Sci. 2012;25(1):26-34.